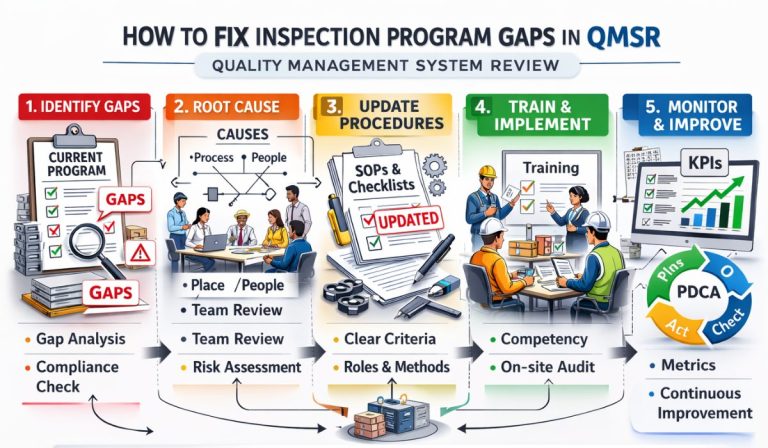
How To Fix Inspection Program Gaps In QMSR
Identifying and closing inspection program gaps is critical for medical device manufacturers transitioning to the FDA’s Quality Management System Regulation (QMSR). This comprehensive guide explores technical strategies, risk-based methodologies, and best practices to ensure your internal audits align with ISO 13485:2016 and FDA expectations, preventing 483 observations and warning letters.